Under this view, equilibrium is described by the following equation:
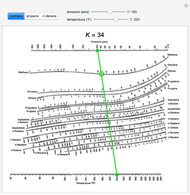
#Depriester chart dew point free#
In other words, the temperature, pressure and molar Gibbs free energy are the same between the two phases when they are at equilibrium.Īn equivalent, more common way to express the vapor-liquid equilibrium condition in a pure system is by using the concept of fugacity.

Where and are the pressures within the liquid and vapor, and are the temperatures within the liquid and vapor, and and are the molar Gibbs free energies (units of energy per amount of substance) within the liquid and vapor, respectively. If the liquid and vapor are pure, in that they consist of only one molecular component and no impurities, then the equilibrium state between the two phases is described by the following equations: Much of the analysis depends on whether the vapor and liquid consist of a single component, of if they are mixtures. The field of thermodynamics describes when vapor-liquid equilibrium is possible, and its properties. Thermodynamic description of vapor-liquid equilibrium A boiling point at an overall pressure of 1 atm is called the normal boiling point. When at a certain temperature, the total of partial pressures of all the components becomes equal to the overall pressure of the system such that vapors generated from the liquid displace any air or other gas which maintained the overall pressure, the mixture is said to boil and the corresponding temperature is the boiling point (This assumes excess pressure is relieved by letting out gases to maintain a desired total pressure). VLE data is often shown at a certain overall pressure, such as 1 atm or whatever pressure a process of interest is conducted at. There can be VLE data for mixtures with even more components, but such data becomes copious and is often hard to show graphically. Three-component mixtures could be called ternary mixtures. A mole fraction is number of moles of a given component in an amount of mixture in a phase (either vapor or liquid phase) divided by the total number of moles of all components in that amount of mixture in that phase.īinary mixtures are those having two components. In mixtures containing two or more components where their concentrations are compared in the vapor and liquid phases, concentrations of each component are often expressed as mole fractions. Distillation takes advantage of differences in concentrations of components in the liquid and vapor phases.

Distillation is a process used to separate or partially separate components in a mixture by boiling (vaporization) followed by condensation. Such VLE information is useful in designing columns for distillation, especially fractional distillation, which is a particular specialty of chemical engineers. In certain cases such VLE data can be determined or approximated with the help of certain theories such as Raoult's Law, Dalton's Law, and/or Henry's Law. Such VLE concentration data is often known or can be determined experimentally for vapor-liquid mixtures with various components. The equilibrium concentration of each component in the liquid phase is often different from its concentration (or vapor pressure) in the vapor phase, but there is a correlation. This fact is true in reverse also if a vapor with components at certain concentrations or partial pressures is in vapor-liquid equilibrium with its liquid, then the component concentrations in the liquid will be set dependent on the vapor concentrations, again also depending on the temperature. At vapor-liquid equilibrium, a liquid with individual components (compounds) in certain concentrations will have an equilibrium vapor in which the concentrations or partial pressures of the vapor components will have certain set values depending on all of the liquid component concentrations and the temperature. The equilibrium vapor pressure of a liquid is usually very dependent on temperature. The concentration of a vapor in contact with its liquid, especially at equilibrium, is often given in terms of vapor pressure, which could be a partial pressure (part of the total gas pressure) if any other gas(es) are present with the vapor.

Vapor-liquid equilibrium, abbreviated as VLE by some, is a condition where a liquid and its vapor (gas phase) are in equilibrium with each other, a condition or state where the rate of evaporation (liquid changing to vapor) equals the rate of condensation (vapor changing to liquid) on a molecular level such that there is no net (overall) vapor-liquid interconversion.
